Bone Marrow Aspiration Device
Bone Marrow Aspiration Device
Aspirate from multiple geographies while limiting peripheral blood dilution — from a single access site, without a centrifuge.

Aspirates freely through the open end — drawing in peripheral blood alongside marrow at a single anatomical point and diluting the cellular yield.
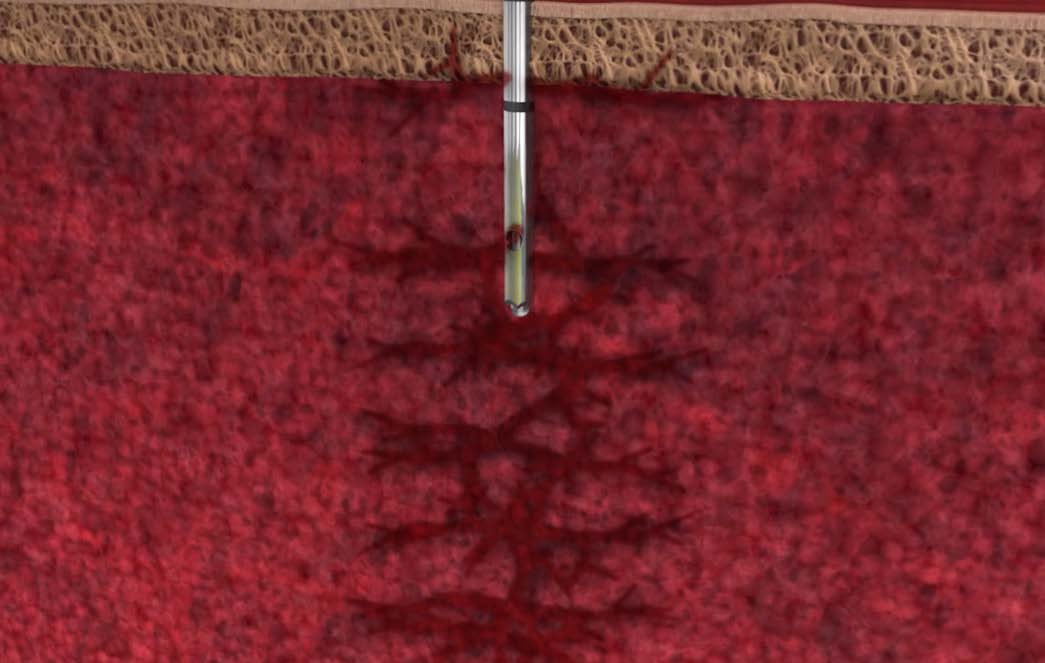
Forces 100% of aspiration through precision side ports — blocking peripheral blood dilution and capturing higher cell counts from undisturbed cancellous bone.
Hover over the device to explore each component — or tap a card below.

CFU-f counts and Total Nucleated Cell density track shoulder-to-shoulder with autograft across four independent studies (n = 37 pooled).
Mesenchymal Stem & Progenitor Cell Density
Total Nucleated Cells
Individual sample results from each study that comprise the Cervos BMA pooled n = 37 cohort.
| Study | Device / System | Sample Size n |
CFU-f per mL |
TNC ×106 per mL |
|---|---|---|---|---|
| Muschler1 | Cancellous Bone Autograft (gold standard) | 10 | 4,564 | 55.7 |
| Lutz5 | Cervos BMA — Marrow Cellution | 22 | 4,838 | 47.0 |
| Scarpone4 | Cervos BMA — Marrow Cellution | 5 | 5,653 | 43.4 |
| Tortland3 | Cervos BMA — Marrow Cellution | 5 | 4,828 | 55.1 |
| Bianco2 | Cervos BMA — Marrow Cellution | 5 | 5,309 | 68.5 |
| Pooled | Cervos BMA — Marrow Cellution | 37 | 5,010 | 50.5 |
Higher CFU-f counts per mL within bone marrow aspirate concentrate have been directly linked to superior clinical outcomes, with research establishing that specific progenitor cell thresholds must be met to achieve meaningful tissue regeneration.6–8
A 2-minute harvest, compared to 20 minutes required when using traditional Bone Marrow Aspiration Concentration (BMAC) systems.
High upfront and ongoing costs: centrifugation systems require significant capital investment, routine maintenance, and dedicated space, increasing total procedural cost beyond the disposables themselves.
Transferring aspirate out of the sterile field for processing introduces additional handling steps, workflow complexity, and potential contamination or re-entry challenges.
The advantages of Cervos (Marrow Cellution) versus spin systems are it is more efficient for the physician and staff, safer from an infection point of view, and it has equal if not superior regenerative properties from a biological standpoint.

Centrifuge-free bone marrow aspiration uses a precision-engineered cannula with a closed distal tip and lateral side ports to draw aspirate exclusively from undisturbed cancellous bone, blocking peripheral blood dilution at the source. The result is a clinically meaningful cell concentration without centrifugation — eliminating the capital equipment, sterile-field handoff, and 15–20 minute off-field processing time of traditional Bone Marrow Aspirate Concentrate (BMAC) workflows. Aspiration to Application® takes under two minutes.
Across four pooled independent studies (n=37), aspirate obtained using the Marrow Cellution™ device produced average CFU-f counts of 5,010 per mL and total nucleated cell density of 50.5 million per mL — within the range of cancellous bone autograft (4,564 CFU-f/mL; 55.7M TNC/mL). The harvest takes approximately two minutes versus the ~20 minutes typical of BMAC centrifugation, eliminates capital equipment requirements, and keeps the entire procedure within a single sterile field.
Sources
A typical bone marrow aspirate harvest using the Marrow Cellution™ device takes approximately two minutes from access to delivery. The device aspirates directly from cancellous bone through precision side ports with no centrifugation step required. Traditional Bone Marrow Aspirate Concentrate workflows generally require 15–20 minutes of off-field processing — the time savings are meaningful in OR settings and procedure scheduling.
Bone marrow aspirate has been investigated across a range of orthopedic and regenerative medicine applications. The Cervos Marrow Cellution™ device specifically has been used in peer-reviewed studies addressing knee osteoarthritis, hip osteoarthritis and labral tears, shoulder osteoarthritis, subchondral bone marrow lesions, spine fusion supplementation, and critical limb ischemia, among other applications. The Cervos resources page lists 17+ published studies. Decisions about clinical use are made by the treating physician based on the patient, the procedure, and the published evidence relevant to the specific application.
Get product updates, clinical evidence, and event news from Cervos Medical.